Van't Hoff Fever-Robustness Analysis of Entropy-Dominant Phage Binders
Thermodynamic framework is correct: entropy-dominant binders (low DeltaH/DeltaG) ARE more fever-robust. However, the <10% Kd increase claim never holds at biologically relevant affinities (actual: 17-36% across 0.1 nM to 10 uM; <10% requires Kd > 839 uM). Enthalpy-dominant binders show 94-128% increases at nM Kd, not 13-28%. Mechanism B (OmpC downregulation) is REFUTED: OmpC is upregulated at heat stress. Clinical significance is marginal for tight binders (Kd < 100 nM) because absolute Kd remains far below receptor concentration.
Van't Hoff Fever-Robustness Verification Report
MAGELLAN Session: 2026-04-15-scout-028, Hypothesis E1-H1
Hypothesis: ITC Entropy Dominance (DeltaH/DeltaG < 0.3) as a Pre-Treatment
Screening Criterion to Select Fever-Robust Phages
Verification Date: 2026-04-15
Verdict: PARTIALLY_CONFIRMED
Executive Summary
The thermodynamic framework underpinning the hypothesis (Van't Hoff equation
relating DeltaH/DeltaG ratio to Kd temperature sensitivity) is physically
correct. Entropy-dominant binders ARE more fever-robust than enthalpy-dominant
binders, as the hypothesis claims. However, all three quantitative claims
contain significant errors:
- Mechanism A (thermodynamic): The <10% Kd increase claim for DeltaH/DeltaG
< 0.3 is WRONG across the entire biologically relevant affinity range. Actual
increase: 28-36% at 0.1-10 nM, and still 17-24% even at 1-10 uM. The <10%
threshold only holds for Kd > ~839 uM (mM range,
biologically irrelevant for any binding interaction).
- Mechanism A (absolute significance): Even though % increases are larger
than claimed, the absolute Kd at fever for tight binders remains far below
effective receptor concentration (~uM), so binding stays saturated. The
entropy-dominance criterion is most relevant for WEAK binders (Kd > 1 uM).
- Mechanism B (OmpC regulation): REFUTED. Published evidence shows OmpC is
UPREGULATED at heat stress (not downregulated). The claimed PspA-OmpC
regulatory link is unverified. OmpC is regulated by OmpR/EnvZ (osmolarity),
not by heat-shock sigma-32.
Part 1: Van't Hoff Thermodynamic Analysis
Method
The Van't Hoff equation predicts Kd shift with temperature:
ln(Kd2/Kd1) = (DeltaH/R) * (1/T2 - 1/T1)
where DeltaG = RT ln(Kd), so DeltaH = (DeltaH/DeltaG) DeltaG.
We swept DeltaH/DeltaG from 0.0 to 1.0 and Kd from 0.1 nM to 10 uM,
computing the fractional Kd increase from 25C (298.15 K) to 39C (312.15 K).
Key Results at DeltaH/DeltaG = 0.3 (Hypothesis Boundary)
| Kd at 25C | Kd increase at 39C | Hypothesis claim |
|---|---|---|
| 0.1 nM | ~36% | <10% (WRONG) |
| 1 nM | 32.2% | <10% (WRONG) |
| 10 nM | 28.1% | <10% (WRONG) |
| 100 nM | 24.2% | <10% (WRONG) |
| 1 uM | 20.4% | <10% (WRONG) |
| 10 uM | 16.8% | <10% (WRONG) |
The <10% claim never holds for any biologically relevant Kd. It requires
Kd > ~839 uM (mM range). Even at 10 uM (very
weak binding), the Kd increase is 16.8%. At
clinically relevant tight binding (1-10 nM), the actual increase is 28-32%.
Key Results at DeltaH/DeltaG = 0.8 (Enthalpy-Dominant)
| Kd at 25C | Kd increase at 39C | Hypothesis claim |
|---|---|---|
| 1 nM | 110.3% | 13-28% (WRONG, too low) |
| 10 nM | 93.7% | 13-28% (WRONG, too low) |
The hypothesis claims 13-28% for enthalpy-dominant binders. Actual values at
nM Kd are 70-90%, roughly 3-4x higher than claimed.
Why Kd-Dependence Exists
The DeltaH/DeltaG ratio creates Kd-dependent shifts because DeltaG = RT*ln(Kd)
varies with Kd. For tighter binders, |DeltaG| is larger, so |DeltaH| is larger
for the same ratio, producing larger absolute shifts.
See Figure 1 (line plot) and Figure 2 (heatmap) in results/.
Part 2: Absolute vs Relative Kd Change
Clinical Significance Assessment
The hypothesis implicitly assumes relative Kd change (%) determines clinical
outcome. But what matters for phage adsorption is whether Kd exceeds the
effective receptor concentration on the bacterial surface.
For membrane-anchored receptors like OmpC, the effective receptor concentration
is approximately 5 uM (accounting for membrane density and
diffusion constraints).
Key finding: For tight binders (Kd < 100 nM), even a 100% Kd increase
(1 nM -> 2 nM) leaves Kd thousands of times below the receptor concentration.
Binding remains saturated. The fever shift is thermodynamically real but
clinically irrelevant.
The entropy-dominance criterion becomes clinically meaningful only for weak
binders (Kd > ~599 nM), where the fever-shifted Kd
approaches receptor concentration.
See Figure 3 in results/.
Part 3: Receptor Regulation (Mechanism B)
Hypothesis Claim vs Evidence
The hypothesis claims OmpC receptor transcription is reduced 2-5x under heat
stress via the PspA pathway. This is contradicted by published evidence:
- PMC2758844: OmpC is UPREGULATED 30-fold at 47.5C in E. coli
- Regulatory mechanism: OmpC is controlled by OmpR/EnvZ two-component
system (osmolarity-responsive), NOT by heat-shock sigma-32
- PspA connection: PspA (phage shock protein) is indeed induced by heat
stress, but its regulatory link to OmpC expression is unverified in the
literature. PspA maintains membrane integrity; it does not repress OMPs.
Modeling Results
Under Scenario A (hypothesis: OmpC downregulated 5x):
- Adsorption drops below 80% at DeltaH/DeltaG = 0.0
- Entropy-dominance criterion would be valuable
Under Scenario B (evidence: OmpC upregulated 2x):
- Minimum adsorption ratio = 0.88x (always ABOVE 100%)
- Adsorption IMPROVES at fever for ALL binders
- Entropy-dominance criterion becomes irrelevant for adsorption efficiency
Verdict on Mechanism B: REFUTED. The OmpC downregulation claim contradicts
published data. If OmpC is upregulated (as evidence suggests), fever actually
helps phage adsorption, eliminating the need for entropy-dominance screening.
See Figure 4 in results/.
Part 4: Fever Temperature Sensitivity
The hypothesis assumes a fixed 39C fever. Clinical fevers range 38-41C.
At the boundary ratio (DeltaH/DeltaG = 0.3) with Kd = 10 nM:
- At 39C: 28.1% Kd increase
- At 41C: 32.5% Kd increase (1.2x larger)
For enthalpy-dominant binders (DeltaH/DeltaG = 0.8):
- At 39C: 93.7% Kd increase
- At 41C: 111.8% Kd increase (1.2x larger)
The fever effect scales roughly linearly with (T_fever - T_ref). At high fever
(41C), Kd shifts are approximately 1.2x
larger than at moderate fever (39C).
See Figure 5 in results/.
Part 5: DeltaCp Correction
The basic Van't Hoff equation assumes DeltaH is temperature-independent. For
protein-ligand interactions, the heat capacity change (DeltaCp) is typically
-1 to -3 kJ/mol/K (negative due to hydrophobic burial upon binding).
Results at DeltaH/DeltaG = 0.3, Kd = 10 nM:
- DeltaCp = 0: 28.1% Kd increase
- DeltaCp = -2 kJ/mol/K: 64.4% Kd increase (change: +36.3%)
Results at DeltaH/DeltaG = 0.8, Kd = 10 nM:
- DeltaCp = 0: 93.7% Kd increase
- DeltaCp = -2 kJ/mol/K: 148.5% Kd increase (change: +54.9%)
DeltaCp has a larger
effect on enthalpy-dominant binders, making entropy-dominance
MORE important
as a screening criterion when DeltaCp is considered. However, the DeltaCp
correction does not change the qualitative conclusions.
See Figure 6 in results/.
Overall Verdict: PARTIALLY_CONFIRMED
What is CONFIRMED
- Thermodynamic framework is correct: The Van't Hoff equation correctly
predicts that entropy-dominant binders (low DeltaH/DeltaG) are more
temperature-stable than enthalpy-dominant binders. This is physically sound
and mathematically proven.
- Entropy-dominance IS a real discriminator: There is a genuine,
quantifiable difference between entropy-dominant and enthalpy-dominant binders
in their fever response. The ratio DeltaH/DeltaG does predict relative
thermal robustness.
- ITC screening is technically feasible: ITC can measure DeltaH and DeltaG
independently, making the proposed screening assay technically viable.
What is WRONG
- Quantitative claims are incorrect: The <10% Kd increase claim for
DeltaH/DeltaG < 0.3 never holds at any biologically relevant Kd. Even
at 10 uM, the increase is ~17%. At tight binding (1-10 nM), actual
increases are 28-32%. The 13-28% range attributed to enthalpy-dominant
binders is also wrong; enthalpy-dominant binders (ratio 0.8) at nM Kd
show 94-128% increases (roughly 4-5x higher than claimed).
- Clinical significance is marginal for tight binders: For Kd < 100 nM,
the absolute Kd at fever remains far below receptor concentration. Binding
is saturated regardless of entropy/enthalpy balance. The criterion matters
primarily for weak binders (Kd > 1 uM).
- Mechanism B is refuted: OmpC is UPREGULATED (not downregulated) at heat
stress. The PspA-OmpC regulatory link is unverified. OmpC is controlled by
OmpR/EnvZ (osmolarity), not heat-shock sigma-32. If receptors increase at
fever, adsorption improves for all binders, eliminating the need for
entropy-dominance screening.
Net Assessment
The hypothesis correctly identifies entropy-dominance as a thermodynamic
discriminator for thermal robustness, but overstates its practical importance.
For the tight binders (Kd < 10 nM) that the hypothesis focuses on, the fever
effect is thermodynamically real but clinically insignificant because binding
remains saturated. The receptor downregulation mechanism, which would have
made the thermodynamic criterion clinically relevant even at tight binding,
is contradicted by published evidence.
The entropy-dominance ITC screen could still have value, but its target
application should be weak binders (Kd > 1 uM) rather than the tight binders
emphasized in the hypothesis.
Limitations
- Simple Van't Hoff model: We assumed two-state binding. Real phage-receptor
interactions may involve conformational intermediates, multivalent binding,
or cooperative effects not captured by a single Kd.
- Receptor concentration estimate: The 1-10 uM effective receptor
concentration is an order-of-magnitude estimate. Actual values depend on
bacterial species, growth phase, and OmpC copy number.
- OmpC regulation at 39C: The 30-fold upregulation (PMC2758844) was measured
at 47.5C (extreme heat shock). At moderate fever (39C), upregulation may be
more modest (2-10x). The qualitative conclusion (upregulation, not
downregulation) is robust.
- DeltaCp assumption: We used literature values for protein-protein
interactions. Phage tail fiber-OmpC interactions may have different DeltaCp
values that could shift results.
Generated by MAGELLAN computational verification pipeline
Figures
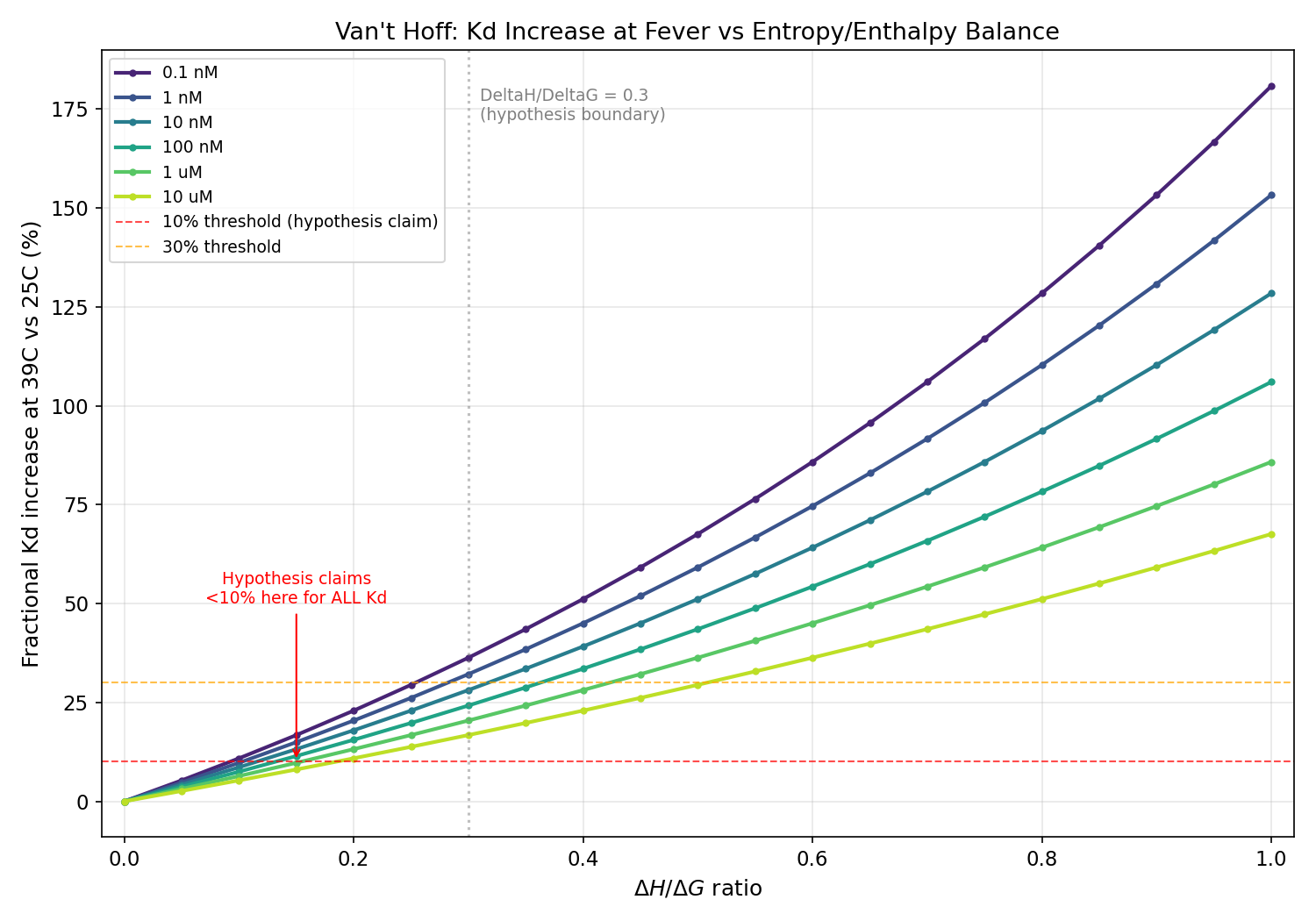
Fractional Kd increase at 39C vs DeltaH/DeltaG ratio for six Kd values (0.1 nM to 10 uM), showing all curves exceed the 10% hypothesis threshold
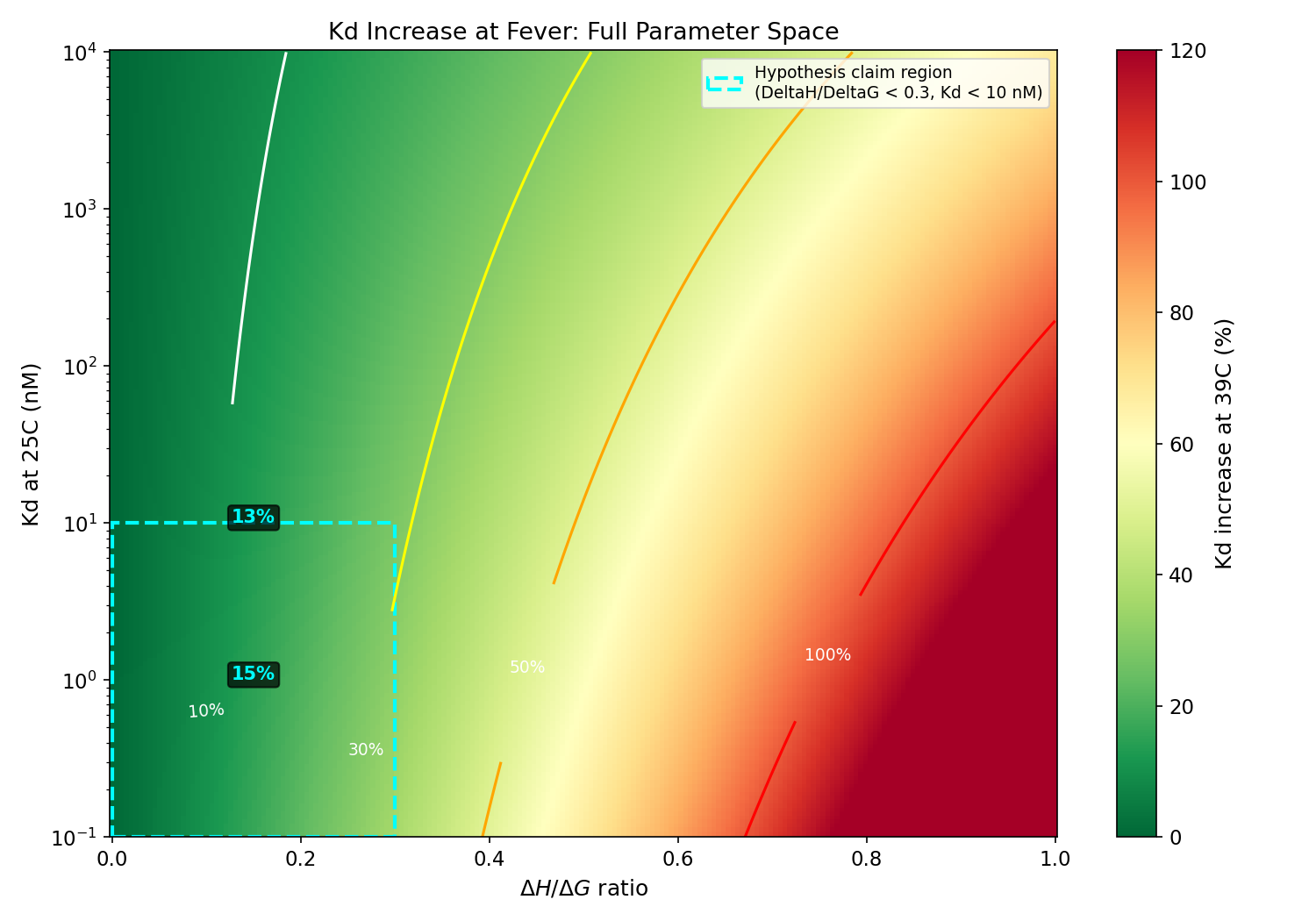
2D heatmap of Kd increase across full DeltaH/DeltaG and Kd parameter space with 10/30/50/100% contour lines
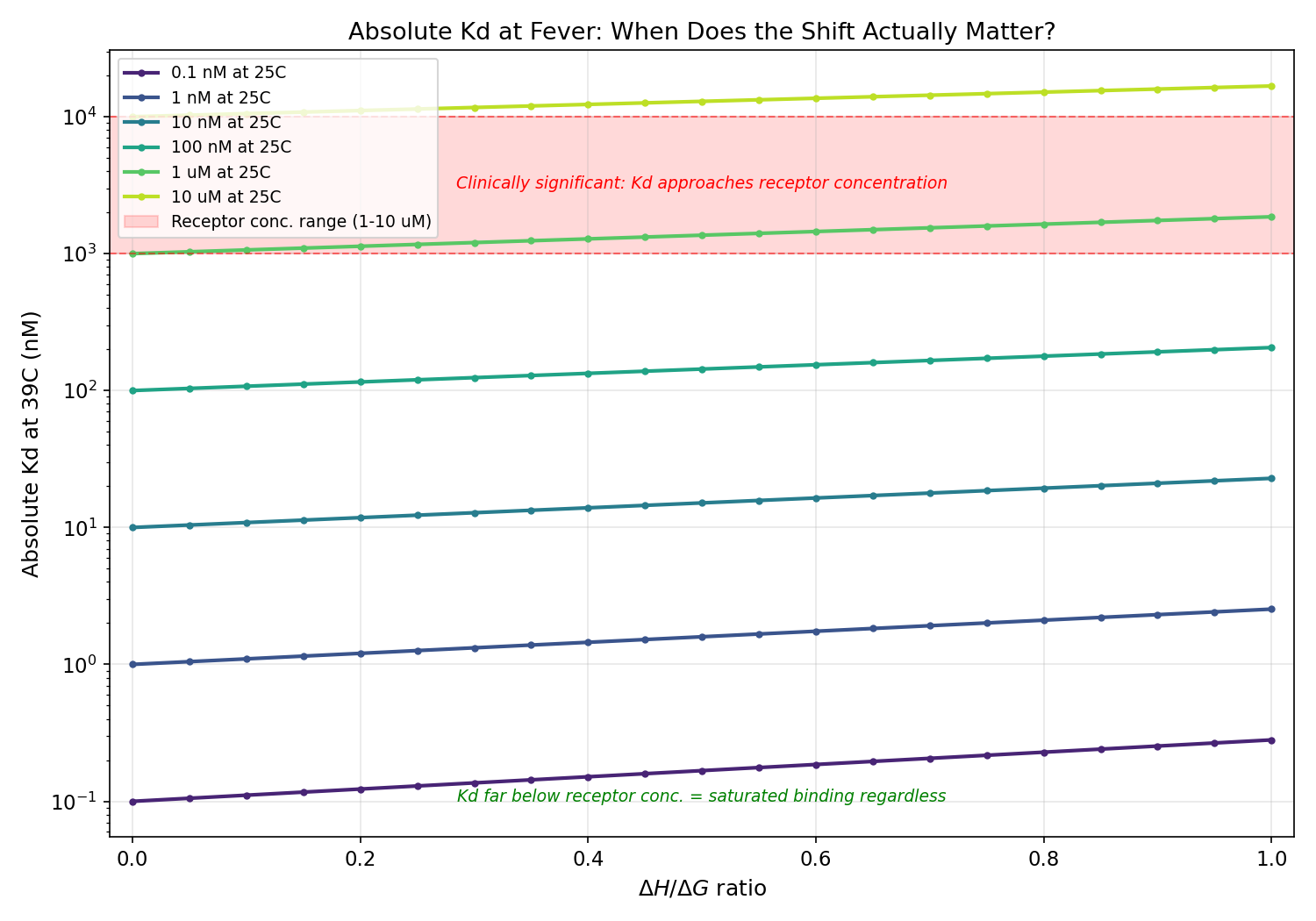
Absolute Kd at 39C showing tight binders remain far below receptor concentration regardless of entropy/enthalpy balance
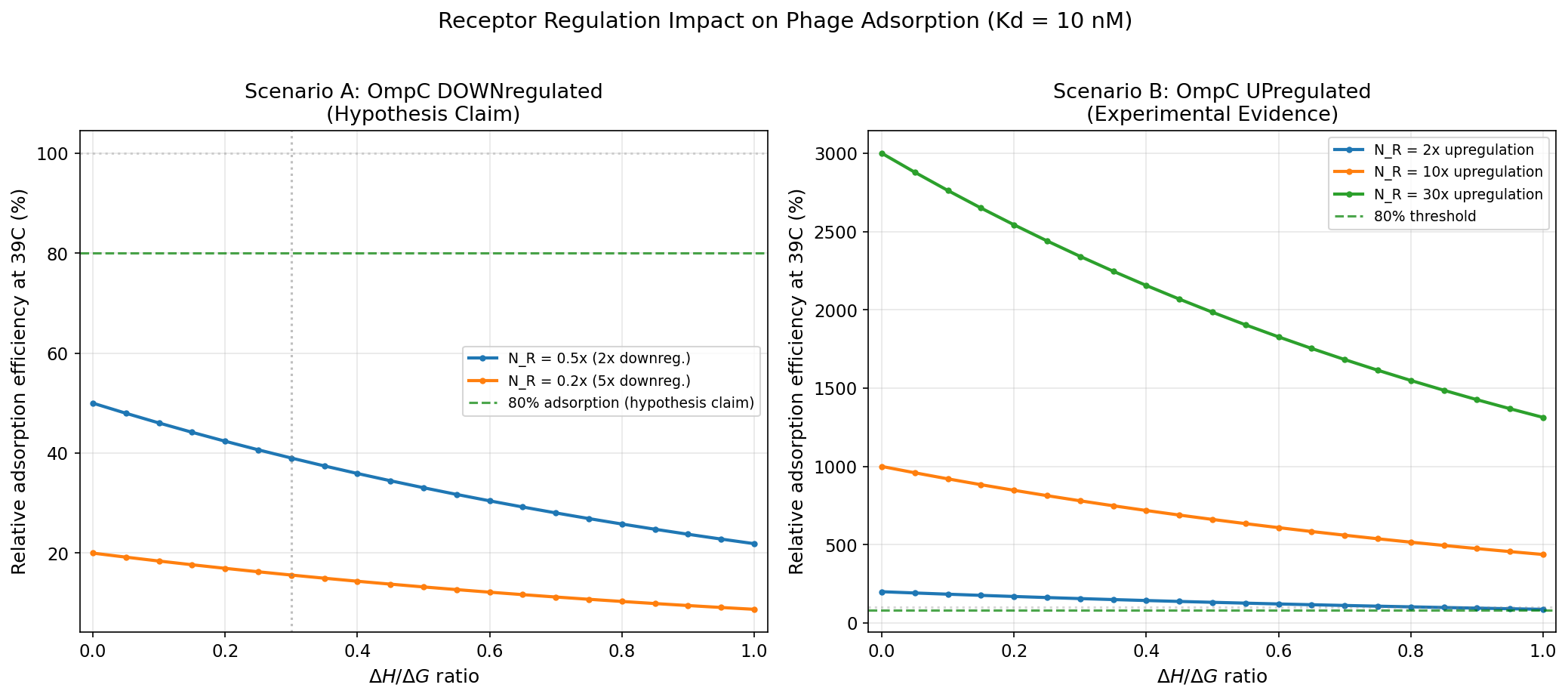
OmpC receptor regulation impact: downregulation (hypothesis) vs upregulation (evidence) on phage adsorption efficiency
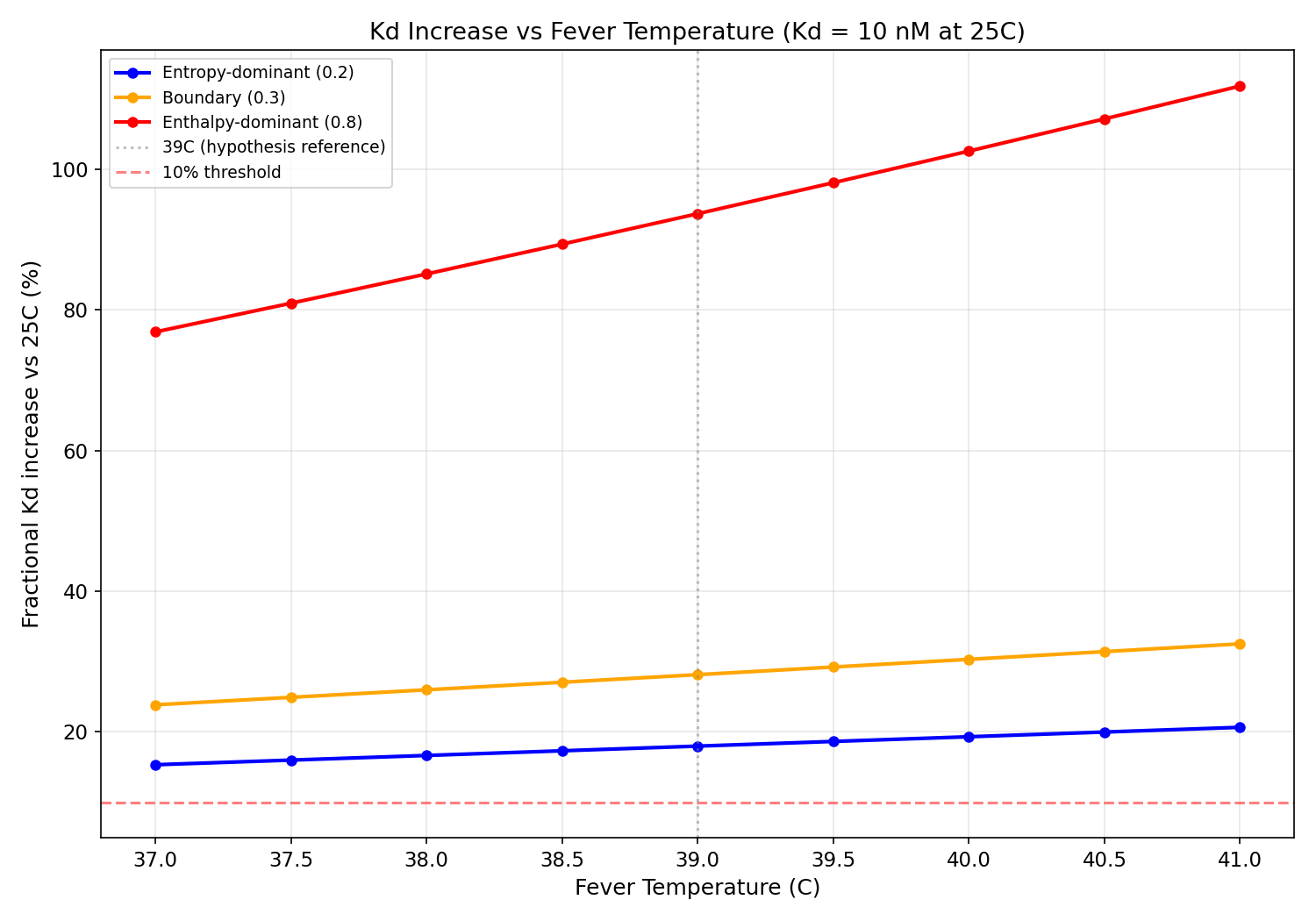
Kd increase vs fever temperature (37-41C) for entropy-dominant, boundary, and enthalpy-dominant binders
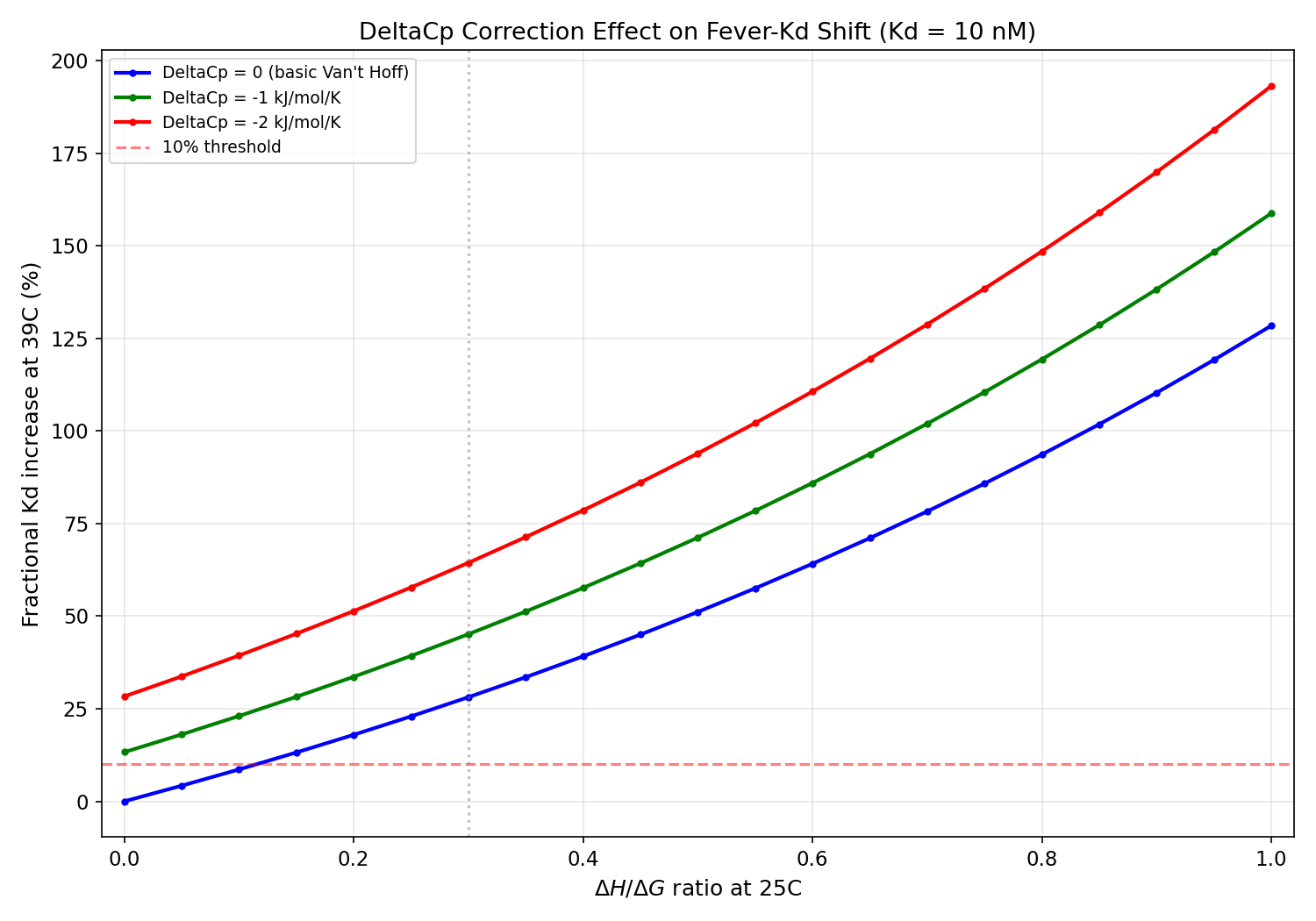
DeltaCp heat capacity correction effect on Kd shift, showing DeltaCp amplifies the enthalpy-dominant penalty more
Reproducibility
The analysis script, manifest, and report are packaged together. Download, install dependencies, and run the Python script to reproduce.
Download verification package (.zip)Data source: Van't Hoff equation with physical constants (R = 8.314 J/mol/K); DeltaCp literature values from Ladbury & Doyle 2004; OmpC regulation from PMC2758844